 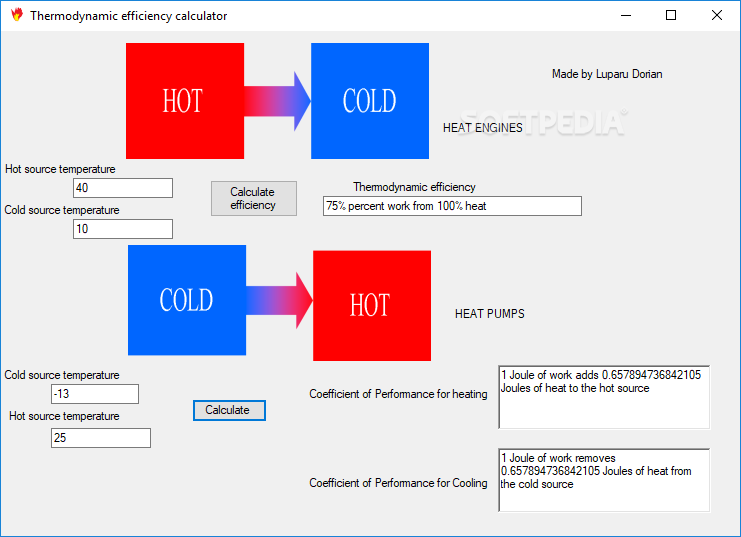 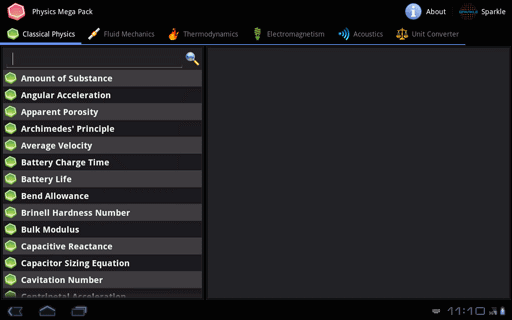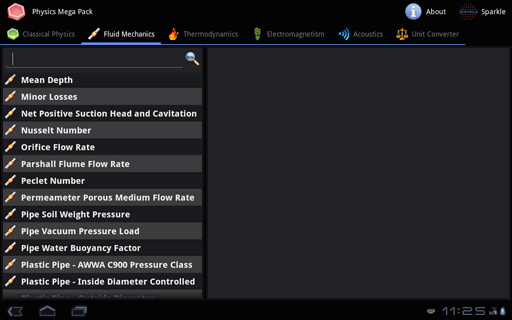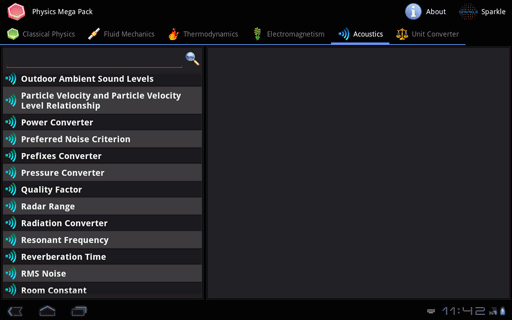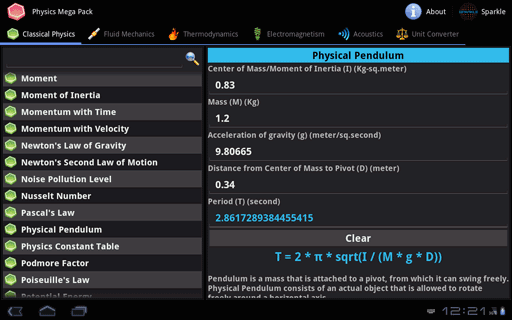
To calculate Work using First Law of Thermodynamics, you need Change in Internal Energy (ΔU) & Heat Transferred in Thermodynamic Process (Q). Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude is calculated using Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process. in surroundings, through suitable passive linkages, work can lift a weight, for example. in thermodynamics, work performed by a system is energy transferred by system to its surroundings, by a mechanism through which system can spontaneously exert macroscopic forces on its surroundings. The Work using First Law of thermodynamics formula is defined as difference between internal energy and heat into system. It is the energy necessary to create or prepare the system in any given internal state & Heat Transferred in Thermodynamic Process is the form of energy that is transferred from the high-temperature system to the low-temperature system. The Change in Internal Energy of a thermodynamic system is the energy contained within it. Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude and is represented as W = ΔU- Q or Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process. 
Here is how the Work using First Law of Thermodynamics calculation can be explained with given input values -> 300 = 900-600. How to calculate Work using First Law of Thermodynamics using this online calculator? To use this online calculator for Work using First Law of Thermodynamics, enter Change in Internal Energy (ΔU) & Heat Transferred in Thermodynamic Process (Q) and hit the calculate button. 
Work done in Thermodynamic Process is denoted by W symbol. Energy can also transfer from surroundings to system in a sign convention used in physics, such work has a negative magnitude. Work using First Law of Thermodynamics calculator uses Work done in Thermodynamic Process = Change in Internal Energy- Heat Transferred in Thermodynamic Process to calculate the Work done in Thermodynamic Process, The Work using First Law of thermodynamics formula is defined as difference between internal energy and heat into system. This option may be re-enabled by the project by placing a file with the name ".How to Calculate Work using First Law of Thermodynamics? NOTE: As of directory index display has been disabled by default. Contact the project administrators of this project via email (see the upper right-hand corner of the Project Summary page for their usernames) at you are a maintainer of this web content, please refer to the Site Documentation regarding web services for further assistance.Contact the project via their designated support resources.If this is a severe or recurring/persistent problem, please do one of the following, and provide the error text (numbered 1 through 7, above): This issue should be reported to the -hosted project (not to ). Reporting this problem: The problem you have encountered is with a project web site hosted by. An error has been encountered in accessing this page. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
